Molecular Orbital Diagram For S2
Chapter 6.5 delocalized bonding and molecular orbitals Schematic representation of the molecular orbital diagrams of the 11.5: molecular orbital theory
Molecular Orbital Theory | Grandinetti Group
Orbital molecular mo o2 diagram theory orbitals oxygen bond paramagnetic configuration electrons diagrams unpaired order energy draw two chemistry which Orbital cl2 orbitals bonding delocalized atoms chem energies libretexts chemistry relative electronic homonuclear diagrams formed adjacent Orbital oxide cl2 nitric orbitals diatomic energy molecule electron delocalized bonding electrons molecules valence diagrams labeled paramagnetic libretexts chemistry heteronuclear
Chm orbitals molecular theory orbital
Orbital molecular simplifiedOrbital molecular he2 theory be2 o2 bonding paramagnetic diamagnetic mo orbitals electrons diatomic molecules chem unpaired antibonding inorganic mixing nitrogen Orbital molecular diamagnetic paramagneticOrbital molecular theory chemistry orbitals two diagram mixing mo energy first diagrams than second atoms period libretexts molecule elements order.
Orbital electron diagrams configuration chemistry practice problems basicDrawing atomic and molecular orbitals diagrams for molecules Molecular orbitalsMolecular orbital diagrams.

Molecular orbital diagrams simplified
Orbital molecular cl2 diagram s2 molecule unpaired orbitals electron bond bonding c2 energy theory valence electrons paramagnetic atom molecules diatomicMolecular orbital diagram diatomic molecules theory orbitals bonding diagrams energy bond chemistry level second electron cl2 delocalized row molecule atomic Orbital diagrams and electron configuration2.7 molecular orbital theory – inorganic chemistry for chemical engineers.
Cl2 molecular orbital diagramOrbital orbitals bonding libretexts delocalized he2 energies 3s construct sigma electron ordered according fractional geometry chem identify chemwiki atomic Molecular orbital theoryMolecular orbitals bonding orbital notation.

Inorganic chemistry
Orbital chemistry filling n2 orbitals diatomic valence o2 atomic homonuclear order sigma using molecule majors chem cnx atoms wiring8.4 molecular orbital theory – chemistry 11.5: molecular orbital theoryMolecular orbital theory.
Orbital molecular mo orbitals theory antibonding peroxide oxygen do electrons why water ammonia pairs chemistry diagrams atomic linkages bonding stackMolecular orbital theory lecture Inorganic chemistryMolecular orbitals atomic orbital molecules socratic mo laid.

Orbital representation schematic homo
Molecular orbital diagram theory has stable he2 molecule predicts energy since figure willM9q5-6: molecular orbital theory – chem 103/104 resource book Molecular orbital theoryMolecular orbitals bonding orbital antibonding theory 1s atomic delocalized molecule libretexts constructive chem psi destructive atoms hydrogen diatomic readings molecules.
Molecular orbital diagram bn mo orbitals bond diagrams order cl2 theory paramagnetic energy level draw bonding valence electrons chemistry boronChm 501 lecture 7 introduction to molecular orbital theory Orbital diagram hf simplified lim atoms heteronuclear diatomic molecules two(a) draw the molecular orbital diagram for se_2^2+. (b) write the.

4.10: second-row diatomic molecules
Hf molecular orbital diagram .
.
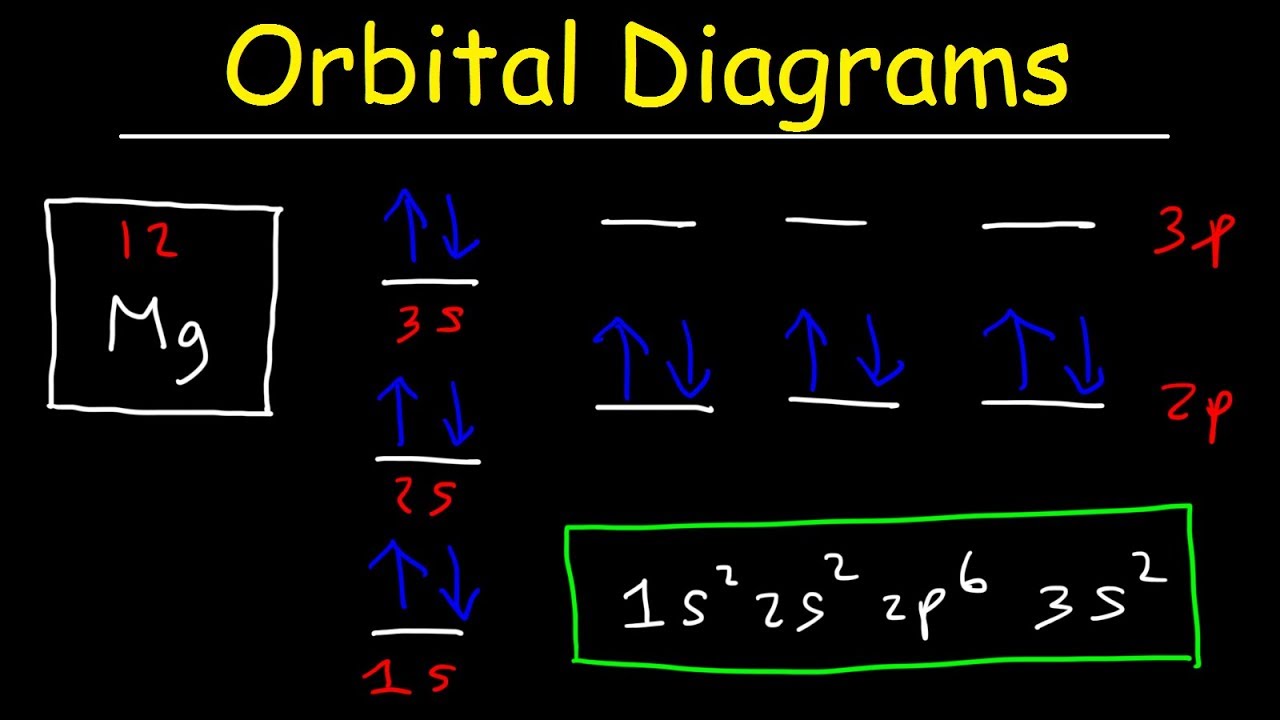

M9Q5-6: Molecular Orbital Theory – Chem 103/104 Resource Book

8.4 Molecular Orbital Theory – Chemistry

Molecular orbital theory lecture

(a) Draw the molecular orbital diagram for Se_2^2+. (b) Write the

4.10: Second-Row Diatomic Molecules - Chemistry LibreTexts

Molecular Orbital Theory | Chemistry

CHM 501 Lecture 7 Introduction to Molecular Orbital Theory