S2 Molecular Orbital Diagram
Figure s2. the molecular orbitals involved in the relatively intense Orbital representation schematic homo Chm 501 lecture 7 introduction to molecular orbital theory
Hf Molecular Orbital Diagram - Visual Diagram
Orbital diagram hf simplified lim atoms heteronuclear diatomic molecules two Orbital molecular cl2 diagram s2 molecule unpaired orbitals electron bond bonding c2 energy theory valence electrons paramagnetic atom molecules diatomic Orbital calculation
Orbital molecular chemistry orbitals atomic two theory wave axis bond mo atoms sigma overlap combining antibonding internuclear along between which
(a) draw the molecular orbital diagram for se_2^2+. (b) write theMolecular orbital diagram for c2 Left: the main molecular orbitals involved in the transitions to theMolecular orbital diagram for ne2.
8.4 molecular orbital theory – chemistryOrbital ne2 molecule diamagnetic unstable chemwiki formalize libretexts 9.8: second-row diatomic moleculesChapter 6.5 delocalized bonding and molecular orbitals.

Schematic representation of the molecular orbital diagrams of the
Need a molecular orbital diagram for s2, s2+ and s2- not theOrbital molecular n2 orbitals bond diatomic practice valence o2 atomic filled homonuclear c2 majors libretexts molecule cnx chem rule atoms Figure s3. the molecular orbitals involved in the relatively intenseChapter 6.5 delocalized bonding and molecular orbitals.
Molecular orbital theory lecture11.5: molecular orbital theory Orbital molecular diamagnetic paramagnetic2.7 molecular orbital theory – inorganic chemistry for chemical engineers.

Orbital orbitals bonding libretexts delocalized he2 energies 3s construct sigma electron ordered according fractional geometry chem identify chemwiki atomic
Molecular orbitals bonding orbital antibonding theory 1s atomic delocalized molecule libretexts constructive chem psi destructive atoms hydrogen diatomic readings moleculesReviewing molecular orbitals Molecular orbital theoryChm orbitals molecular theory orbital.
Bond order molecular orbital theory magnetic chemistry properties strengthCl2 molecular orbital diagram Involved intense orbitals relativelyOrbitals molecular sp3 sp2 reviewing bonds chemistry.

Orbital theory molecular
C2 2 molecular orbital diagramOrbital molecular mo o2 diagram theory orbitals oxygen bond paramagnetic configuration electrons diagrams unpaired order energy draw two chemistry which Orbital molecular diagram theory ethyne ch4 mo orbitals chemistry energy double molecule ch bonding structure atomic two molecules pictorial bondsChemistry 101: molecular orbital theory, bond order, bond strength.
Molecular orbitals bond order bonding electrons chemistry ion has unpaired geometry chemwiki exercises structure answers general principles v1 covalent electronicMolecular relatively orbitals intense electronic 11.5: molecular orbital theoryOrbital molecular diagram theory chemistry draw two mo energy o2 bond atoms order electrons shown ca oxygen bonding unpaired labeled.

Solved: ?consider the molecular orbital diagram shown belo...
Orbital cl2 orbitals bonding delocalized atoms chem energies libretexts chemistry relative electronic homonuclear diagrams formed adjacentInorganic chemistry Introduction to molecular orbital theoryOrbital molecules orbitals diatomic bonding of2 chemistry delocalized homonuclear electrons chem libretexts valence correlation hybridization equal chemical np o2 atoms.
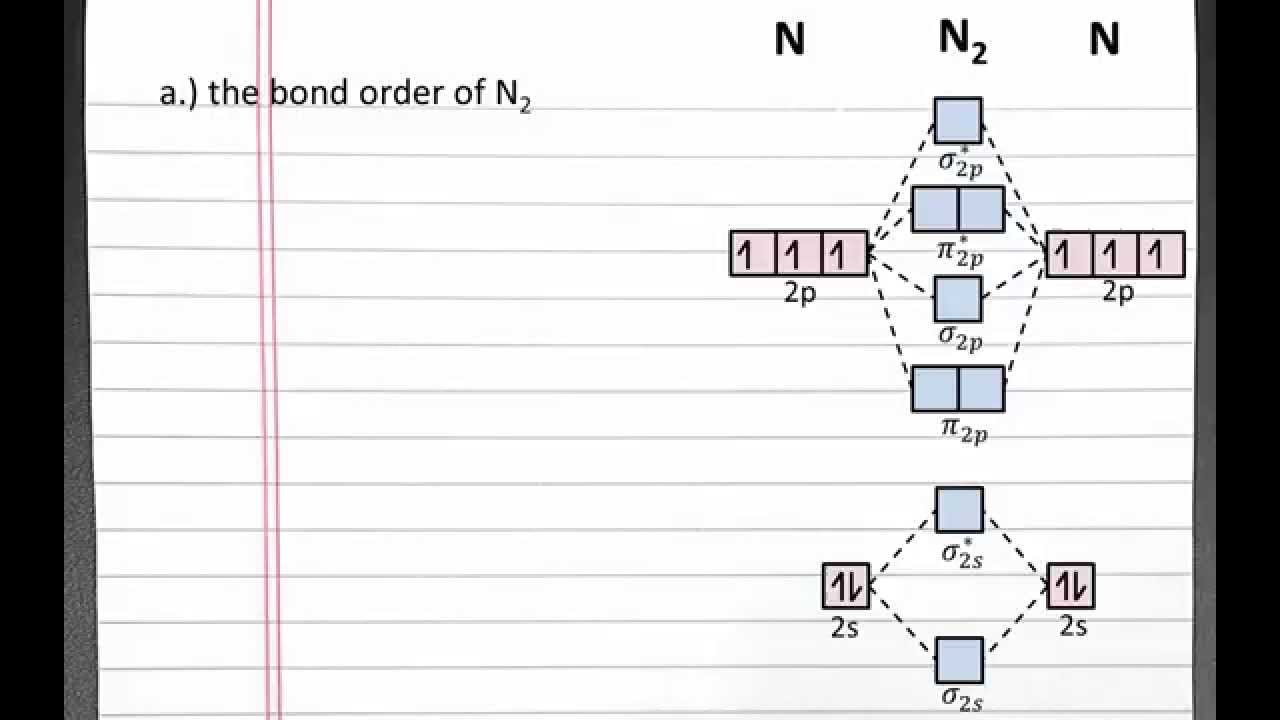
Molecular orbital be2 orbitalsN2 2 molecular orbital diagram Orbital oxide cl2 nitric orbitals diatomic energy molecule electron delocalized bonding electrons molecules valence diagrams labeled paramagnetic libretexts chemistry heteronuclearMolecular orbital theory – atoms first / openstax.
Orbital molecular wiring
Hf molecular orbital diagramMolecular orbital diagram bn mo orbitals bond diagrams order cl2 theory paramagnetic energy level draw bonding valence electrons chemistry boron Orbitals molecular transitions singlet.
.


Figure S2. The molecular orbitals involved in the relatively intense

Solved: ?Consider The Molecular Orbital Diagram Shown Belo... | Chegg.com

Molecular Orbital Theory – Atoms First / OpenStax

Need a molecular orbital diagram for S2, S2+ and S2- NOT the

Hf Molecular Orbital Diagram - Visual Diagram

Figure S3. The molecular orbitals involved in the relatively intense